Heating and acidification can produce toxic chlorine gas. However, it can react explosively with numerous chemicals, including urea, ammonium salts, methanol, acetylene, and many organic compounds. Potassium hypochlorite is not considered a fire or explosive hazard by itself. Ingestion of strong concentrations can be lethal. Inhalation of a mist of KClO can cause bronchial irritation, difficulty breathing, and in severe cases pulmonary edema. It can cause severe damage on contact with the skin, eyes, and mucous membranes. Like sodium hypochlorite, potassium hypochlorite is an irritant. Due to production difficulties, the product was then modified using sodium instead of potassium, giving rise to sodium hypochlorite, widely used today as a disinfectant. The resulting liquid, known as " Eau de Javel" ("Javel water"), was a weak solution of potassium hypochlorite. Potassium hypochlorite was first produced in 1789 by Claude Louis Berthollet in his laboratory located in Javel in Paris, France, by passing chlorine gas through a solution of potash lye. Because its degradation leaves behind potassium chloride rather than sodium chloride, its use has been promoted in agriculture, where addition of potassium to soil is desired. Potassium hypochlorite is used for sanitizing surfaces as well as disinfecting drinking water. With both methods, the reaction mixture must be kept cold to prevent formation of potassium chlorate. Redox equations that need to be balanced can often be written without water molecules, H+ and OH- ions. Īnother production method is electrolysis of potassium chloride solution. This is the traditional method, first used by Claude Louis Berthollet in 1789. Potassium hypochlorite is produced by the disproportionation reaction of chlorine with a solution of potassium hydroxide: Cl 2 + 2 KOH → KCl + KClO + H 2O frequency of Cl 35 in KClO 3 at 273.16 °K and atmospheric pressure is 28,213,372 ☒ Hz. It has a light grey color and a strong chlorine smell. It is used in variable concentrations, often diluted in water solution. Some companies have suggested its application as a substitute for NaOCl as a bleaching agent, alleging the beneficial properties of the K+ ion against the environmental impact caused by Na+.Potassium hypochlorite (chemical formula KClO) is the potassium salt of hypochlorous acid. The degradation in soils of potassium hypochlorite into potassium chloride has suggested its use in crops as a source of potassium, the primary nutritional element for plants. It is used as a disinfectant for surfaces and drinking water. Potassium hypochlorite must be kept cold to avoid the formation of potassium chlorate, whose decomposition can even be explosive. It decomposes, releasing oxygen, a process that can be catalyzed by rust or by the metal container that contains it. Its reaction with nitromethane, methanol and ethanol may become explosive. Likewise, its reaction with organic matter, oil, hydrocarbons and alcohols can cause explosions. It combines with acetylene to form the explosive chloroacetylene. Reacts vigorously with carbon in a potentially explosive reaction. When heated or in contact with acids, it produces highly toxic chloride fumes. In contact with urea it can form NCl3, a highly explosive compound. However, it is capable of combining with different chemical elements to create flammable and explosive compounds. Potassium Hypochlorite is a powerful oxidizing agent and is not considered a fire or explosion hazard. Likewise, its inhalation causes bronchial irritation, respiratory distress and pulmonary edema. 
It is irritating and can cause severe damage in contact with the skin, eyes and mucous membranes. 
The average oxidation state of the 1 K atom in KClO 3 is +1. 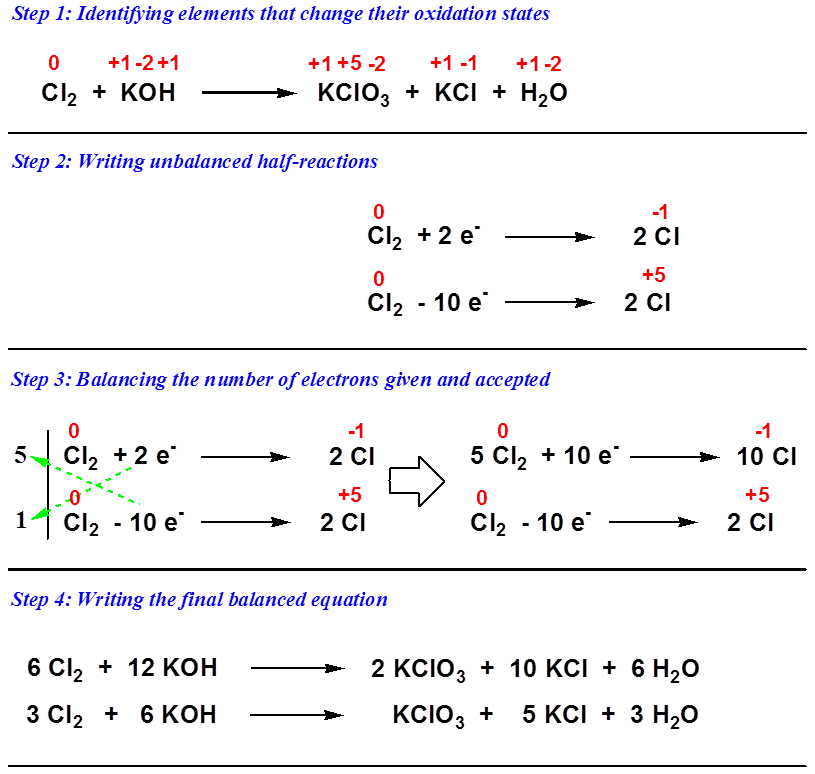
Aqueous solutions of potassium hypochlorite have bleaching qualities, just like those of NaOCl. Cl K Charge From Assigned Electrons-5: 5: 0: Ionic Charge-1: 0: 1: Total Charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
